How Red Light Therapy Affects Cells, Mitochondria, and the Body
Red light - what is it
Red light therapy, also known as photobiomodulation (PBM) or low-level light therapy (LLLT), is increasingly used in physical therapy, rehabilitation, tissue regeneration, and anti-aging treatments. But how can something as simple as light have such powerful effects on the human body?
The answer lies in how certain wavelengths of light—especially red (620–670 nm) and near-infrared (760–850 nm)—interact with the fundamental parts of our cells, particularly mitochondria.
Mitochondria – The Cell’s “Power Plant”
Mitochondria are tiny structures inside our cells, often called the “batteries” or “power plants” of the cell. They produce ATP (adenosine triphosphate)—the primary energy molecule that powers almost every cellular function. Without ATP, cells can’t perform properly, leading to fatigue, poor tissue healing, chronic inflammation, and other issues.
A key enzyme in the production of ATP is cytochrome c oxidase (CcO), which sits at the end of the mitochondrial respiratory chain and enables electron transfer and energy production.
How Does Red Light Work?
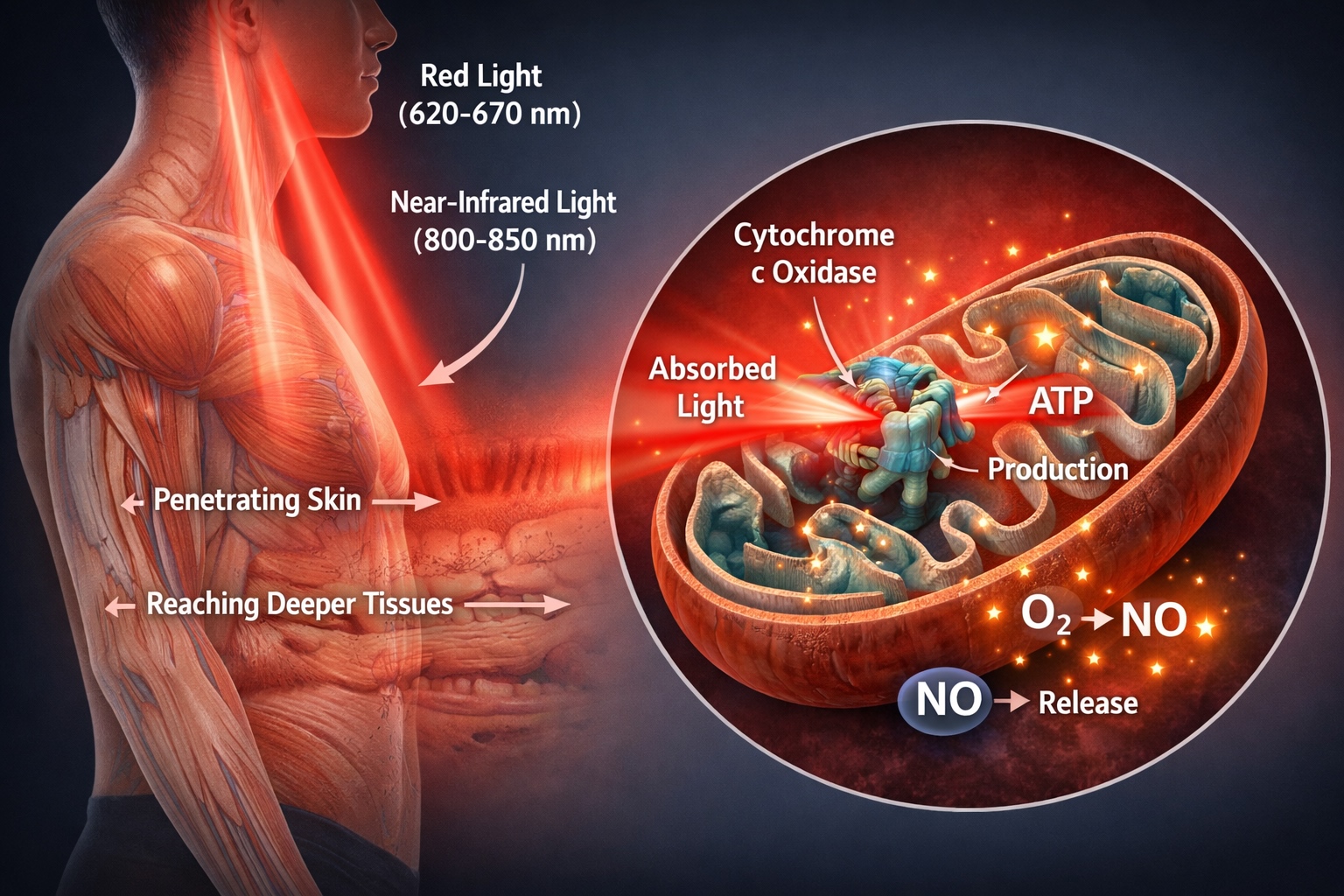
Red and near-infrared light penetrate the skin and reach deeper tissues. Once inside the body, the light is absorbed by mitochondria, specifically by cytochrome c oxidase. This triggers several beneficial biological processes:
How Does Red Light Work?
Red and near-infrared light penetrate the skin and reach deeper tissues. Once inside the body, the light is absorbed by mitochondria, specifically by cytochrome c oxidase. This triggers several beneficial biological processes:
Increased ATP Production
Light exposure stimulates cytochrome activation, enhancing the flow of electrons in the mitochondrial respiratory chain. This leads to greater ATP production, giving the cell more energy for repair, defense, and proper function.
Displacement of Nitric Oxide (NO)
Under stress or inflammation, nitric oxide can bind to CcO and block oxygen, inhibiting ATP production. Red light helps to displace NO, allowing normal oxygen flow and energy generation to resume.
Controlled Reactive Oxygen Species (ROS) Signaling
A small, controlled increase in ROS induced by light is not harmful; rather, it acts as a signal for cellular repair. This activates genes responsible for healing, anti-inflammatory responses, and new blood vessel formation.
Reduction in Inflammation and Pain
PBM reduces pro-inflammatory cytokines like TNF-α and IL-6, while increasing IL-10, a powerful anti-inflammatory. It also modulates nerve conduction and reduces the sensitivity of pain receptors, helping to relieve pain without medication.
Improved Blood Flow and Oxygenation
Red light stimulates angiogenesis (the creation of new capillaries) and enhances endothelial function, increasing oxygen and nutrient delivery to injured tissues.
Tissue Repair and Regeneration
Light stimulation promotes the growth of fibroblasts—the cells responsible for producing collagen—and accelerates the healing of tendons, muscles, skin, and even bones. PBM is linked to faster recovery after sports injuries, surgery, and chronic pain conditions.
Does Wavelength Matter?
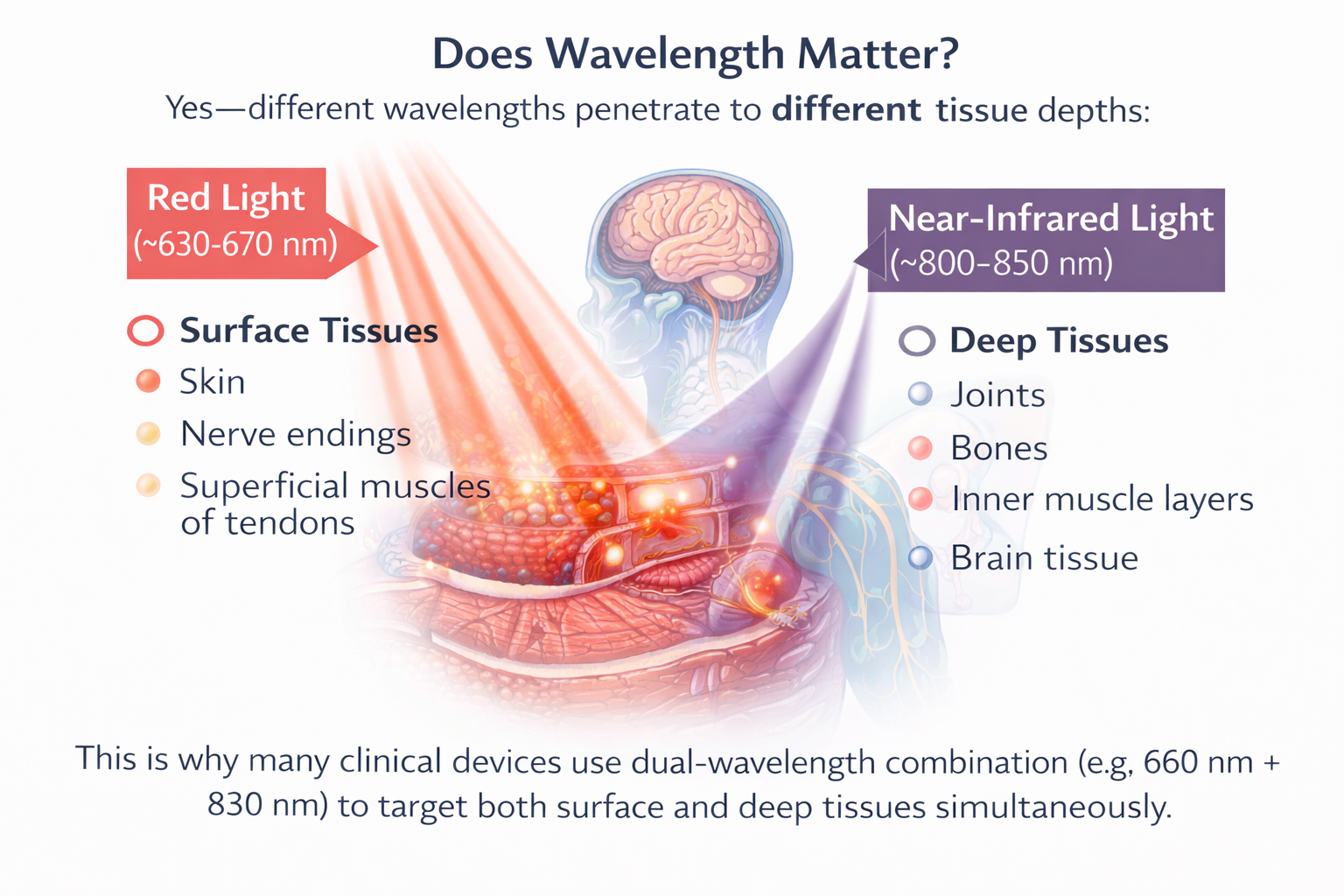
Yes—different wavelengths penetrate to different tissue depths:
Red light (~630–670 nm): ideal for surface tissues—skin, nerve endings, and superficial muscles or tendons.
Near-infrared light (~800–850 nm): penetrates deeper—to joints, bones, inner muscle layers, and even brain tissue.
This is why many clinical devices use dual-wavelength combinations (e.g., 660 nm + 830 nm) to target both surface and deep tissues simultaneously.
Benefits for Muscles and Tendons
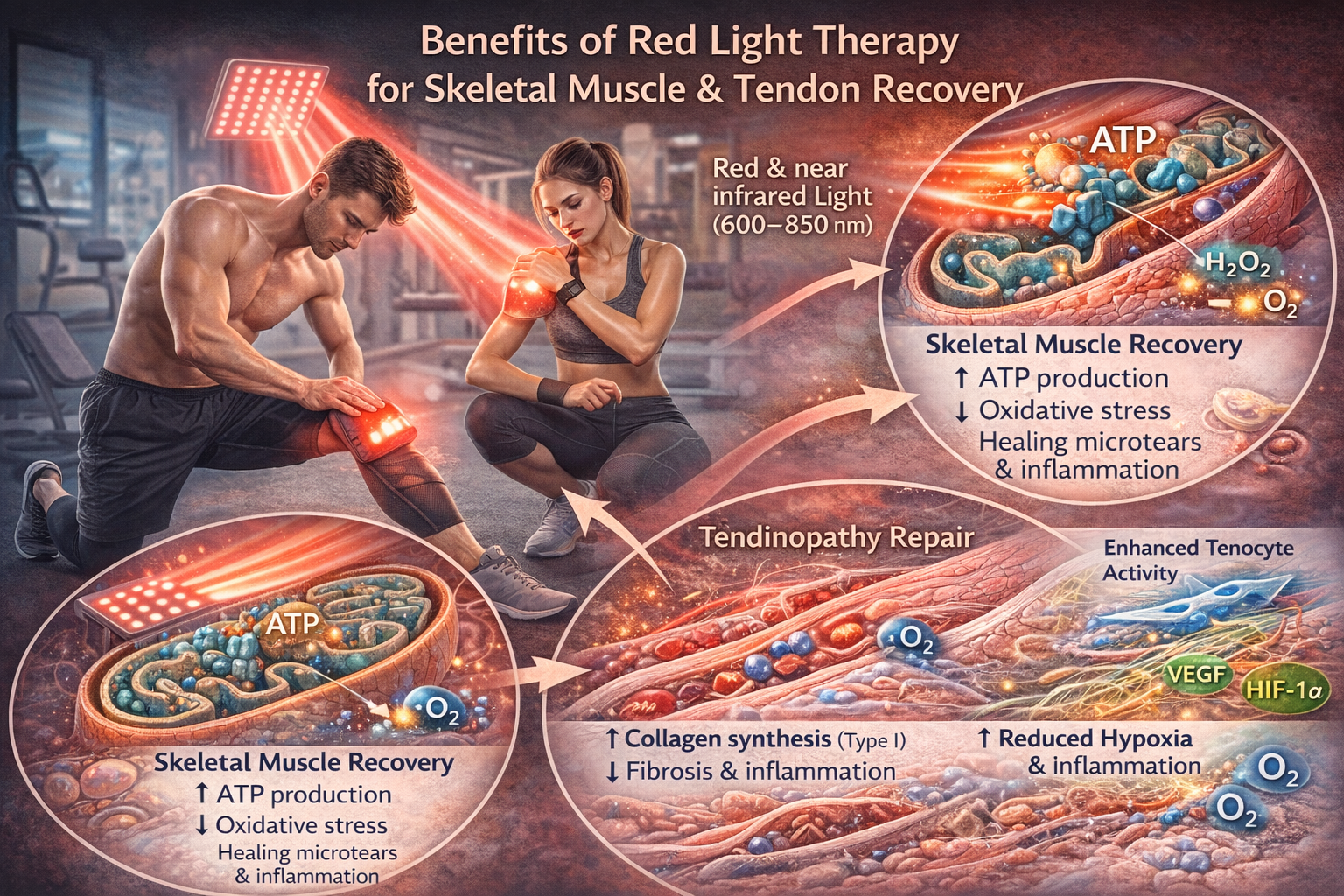
Red light therapy has shown clear benefits for skeletal muscle and tendon recovery. Muscle tissue is highly metabolic and contains abundant mitochondria, making it particularly responsive to photobiomodulation.
When applied to muscles, red and near-infrared light increases ATP production, reduces exercise-induced oxidative stress, and promotes faster repair of microtears and inflammation. Studies have shown that red light therapy can help reduce delayed onset muscle soreness (DOMS), enhance strength recovery, and improve endurance when applied before or after intense activity.
In tendons, red light promotes the activity of tenocytes (tendon cells), enhancing collagen type I synthesis, organization of extracellular matrix, and mechanical strength of the tissue. It also helps to reduce chronic tendon inflammation by downregulating VEGF and HIF-1α expression, factors associated with tissue hypoxia and disorganized angiogenesis.
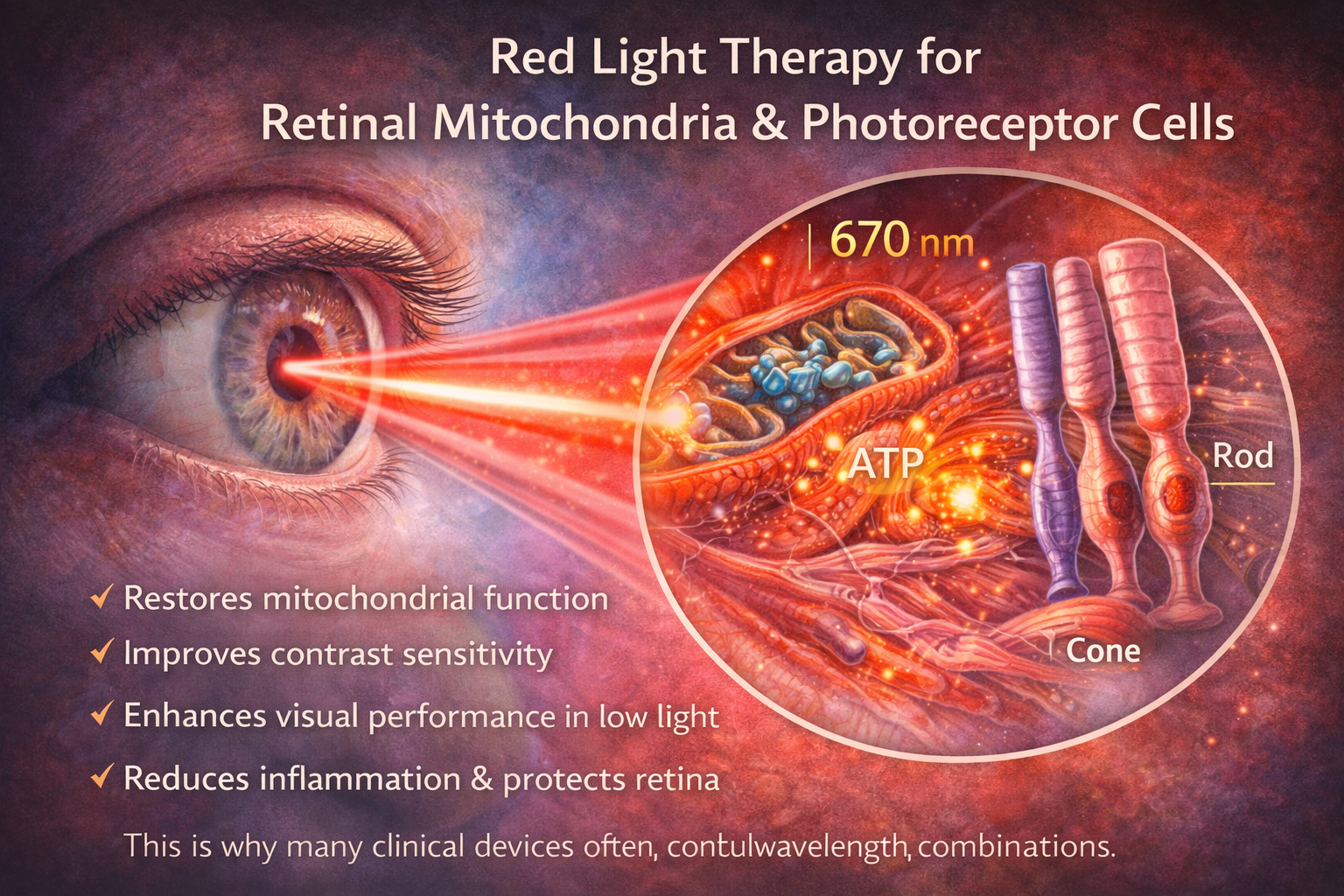
Benefits for Eye Health
Recent studies show that red light therapy may have positive effects on retinal mitochondria and photoreceptor cells, especially in aging populations. As we age, mitochondrial efficiency in the retina declines, leading to reduced visual performance, slower dark adaptation, and increased risk of degenerative diseases.
Exposing the eye to red light has been shown to restore mitochondrial function in retinal cells, improve contrast sensitivity, and enhance overall visual performance, especially in low-light conditions. These benefits appear most pronounced when applied in the morning and for short durations.
Additionally, red light therapy may help reduce inflammation in ocular tissues and support the repair of damaged retinal cells without adverse effects, provided that proper light intensity and exposure time are respected.
References
1. Karu, T. I. (1999). Primary and secondary mechanisms of action of visible to near-infrared radiation on cells. Journal of Photochemistry and Photobiology B: Biology, 49(1), 1–17.
2. Hamblin, M. R., & Demidova, T. N. (2006). Mechanisms of low level light therapy. Proceedings of SPIE, 6140, 614001.
3. de Freitas, L. F., & Hamblin, M. R. (2016). Photobiomodulation and the brain: Effects on mitochondrial function, neuroinflammation, and behavior. Biochimica et Biophysica Acta (BBA) – Bioenergetics, 1859(8), 911–918.
4. Wong-Riley, M. T. T., Liang, H. L., Eells, J. T., Chance, B., Henry, M. M., Buchmann, E., & Kane, M. (2005). Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: Role of cytochrome c oxidase. Annals of the New York Academy of Sciences, 1053, 210–221.
5. Hamblin, M. R. (2017). Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. AIMS Biophysics, 4(3), 337–361.
6. Karu, T. I., Pyatibrat, L. V., & Afanasyeva, N. I. (2004). Cellular effects of low power laser therapy can be mediated by nitric oxide. Lasers in Surgery and Medicine, 34(4), 307–314.
7. Poyton, R. O., & Ball, K. A. (2011). Therapeutic photobiomodulation: Nitric oxide and cytochrome c oxidase. Mitochondrion, 11(3), 523–534.
8. Ferraresi, C., Hamblin, M. R., & Parizotto, N. A. (2012). Low-level laser (light) therapy (LLLT) on muscle tissue: Performance, fatigue and repair benefited by the power of light. Photochemistry and Photobiology, 88(3), 687–700.
9. Leal Junior, E. C. P., Vanin, A. A., Miranda, E. F., de Carvalho, P. D. T. C., Dal Corso, S., & Bjordal, J. M. (2015). Effect of phototherapy (low-level laser therapy and light-emitting diode therapy) on exercise performance and markers of exercise recovery: A systematic review with meta-analysis. Lasers in Medical Science, 30(2), 925–939.
10. Alves, A. N., Fernandes, K. P. S., Deana, A. M., Bussadori, S. K., & Mesquita-Ferrari, R. A. (2014). Effects of low-level laser therapy on skeletal muscle repair: A systematic review. American Journal of Physical Medicine & Rehabilitation, 93(12), 1073–1085.
11. Tumilty, S., Munn, J., McDonough, S., Hurley, D. A., Basford, J. R., & Baxter, G. D. (2010). Low level laser treatment of tendinopathy: A systematic review with meta-analysis. Photomedicine and Laser Surgery, 28(1), 3–16.
12. Bjordal, J. M., Lopes-Martins, R. Á. B., & Iversen, V. V. (2006). A randomized, placebo-controlled trial of low level laser therapy for activated Achilles tendinitis. British Journal of Sports Medicine, 40(1), 76–80.
13. Wu, Y., Wang, J., Gong, Y., & Zhang, L. (2023). Photobiomodulation promotes collagen remodeling and tenocyte activity in tendon healing. International Journal of Molecular Sciences, 26(5), 2286.
14. Jeffery, G., Shinhmar, H., Chaine, G., & Hogg, C. (2020). Mitochondrial function is improved in the aged retina following exposure to 670 nm light. The Journals of Gerontology: Series A, 75(11), 2064–2073.
15. Gkotsi, D., Begum, R., Salt, T., Lascaratos, G., Hogg, C., Chau, K. Y., Schapira, A. H. V., & Jeffery, G. (2014). Recharging mitochondrial batteries in old eyes: Near-infrared increases ATP. Neurobiology of Aging, 35(8), 1870–1873.
16. Calderhead, R. G. (2017). The photobiological basics behind light-emitting diode (LED) phototherapy. Laser Therapy, 26(2), 77–89.
17. Anders, J. J., Lanzafame, R. J., & Arany, P. R. (2015). Low-level light/laser therapy versus photobiomodulation therapy. Photomedicine and Laser Surgery, 33(4), 183–184.
18. Huang, Y. Y., Chen, A. C. H., Carroll, J. D., & Hamblin, M. R. (2009). Biphasic dose response in low level light therapy. Dose-Response, 7(4), 358–383.
